 
This is important as it is the Valence electrons 3d5 4s1, electrons in the outermost shell that determine the chemical properties of the element.Unabbreviated electronic configuration of neutral.  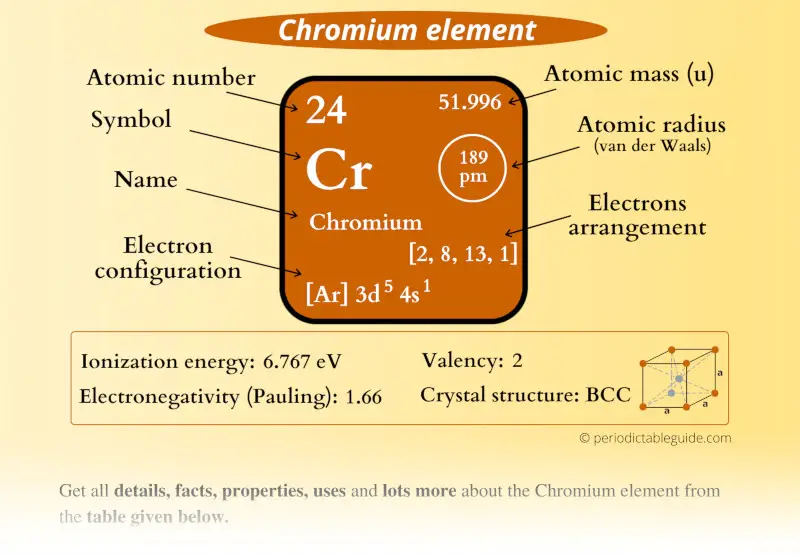
Chromium is a lustrous silver-colored metal that has been used to prevent. For atoms with many electrons, this notation can become lengthy and so an abbreviated notation is used. Therefore, the (4s)^1 do not contribute since we are focusing on the 3d orbital. Which of the following would have the same number of valence electrons. Electrons in shells higher than the electron for which you're solving do not contribute to shielding. * Electrons in a "d" or "f" orbital in the same shell as the electron for which you're calculating contribute 0.35, and electrons in an "d" or "f" orbital in all lower energy levels contribute 1. Valence electrons are responsible for most of the chemical bonding. Electrons in the outermost energy level of an atom are known as valence electrons. Chromium is a hard, lustrous steel-gray metal that was discovered in 1797. Now we will learn how to determine the valence electrons of niobium. However, the valence electron of the transition element can be easily determined according to the Aufbau principle. S = (5 x 0.35) + (8 x 0.85) + (10 x 1) = 18.55 Become a member to unlock this answer Create your account. This is because the valence electrons of the transition elements are located in the inner shell. Therefore, the (4s)^1 do not contribute since we are focusing on the 3d orbital. Metals tend to have high melting points and boiling points suggesting strong bonds between the atoms. A sheet of aluminum foil and a copper wire are both places where you can see metallic bonding in action. *Electrons in an "s" or "p" orbit in the same shell as the electron for which you're solving contribute 0.35, electrons in an "s" or "p" orbital in the shell one energy level lower contribute 0.85, and electrons in an "s" or "p" orbital in shells two energy levels and lower contribute 1. Whereas ionic bonds join metals to non-metals, metallic bonding joins a bulk of metal atoms. Let us first calculate S using Slater's rule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
